Mimicking nature’s protection to fight post-weaning diarrhea from the start
Written by Sofie Tanghe, Researcher Specialties
Ever since the 2022 EU ban on the use of pharmacological levels of zinc oxide in piglet diets, and the reduction in antibiotic use to halt antimicrobial resistance spreading, nutritional measures to prevent and reduce post-weaning diarrhea in piglets have been playing an important role. Several feed-related measures – including feed formulation, feed ingredients, feed additives, and feeding strategies – need to be combined in order to combat post-weaning diarrhea successfully. One of these measures, taken by Earlyfeed experts, is the use of START+: an innovative prebiotic concept based on components that naturally occur in sow milk.
Stimulating early gut maturation
Earlyfeed’s START+ concept contains galacto-oligosaccharides (GOS) that mimic the prebiotic oligosaccharides that are naturally present in sow colostrum and milk. Studies conducted by Earlyfeed’s researchers have shown that including START+ in piglet creep feed stimulates the early maturation of the gastro-intestinal tract of newborn piglets (Tanghe et al., 2020). Several trials have demonstrated longer villi and better villus height/crypt depth ratio, a more beneficial microbiome, and an increased production of short chain fatty acids. This improved early gut maturation prepares the piglet ideally for the stressful event of weaning. Moreover, new research has now revealed that START+ also plays an important protective role in post-weaning diarrhea.
Combating post-weaning diarrhea
Post-weaning diarrhea – which is often caused by enterotoxigenic Escherichia coli F4 (ETEC F4) infection – is a significant economic threat to the global swine industry. To colonize the small intestine, ETEC F4 uses its F4 fimbriae or adhesins to adhere to host-specific receptors present on the enterocytes of the small intestine. Once ETEC F4 is attached and has colonized the small intestine, it produces enterotoxins that lead to diarrhea (Luise et al., 2019).
Thanks to their prebiotic function, GOS can indirectly prevent pathogen infections, because a beneficial gut microbiome can inhibit the growth of pathogens via competitive exclusion. Furthermore, a recent peer-reviewed article – published in Frontiers in Veterinary Science – provides evidence that GOS can also have direct microbiota-independent protective effects against ETEC F4 infection (Guantario et al., 2026). This study was a collaboration between the R&D team of Royal Agrifirm Group and two research groups from CREA (Italy) and Ghent University (Belgium).
Reducing the adhesion of ETEC F4
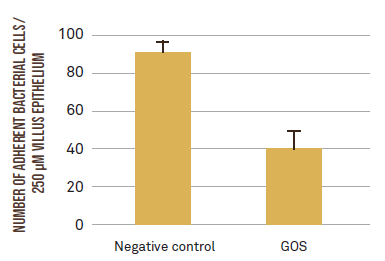
First of all, the research team investigated whether GOS could prevent ETEC F4 adhesion to the intestinal cells in an in vitro intestinal cell model, using the epithelial Caco-2 cell line. GOS structurally resemble the receptors on the intestinal epithelial cells to which intestinal pathogens adhere. Therefore, GOS act as decoy receptors (or anti-adhesives) that competitively inhibit pathogen adherence and subsequent infection. This study has demonstrated that GOS can significantly reduce the adhesion of ETEC F4 to the Caco-2 intestinal cells (6.5 vs 5.9 log CFU/mL, for ETEC vs ETEC+GOS; P < 0.001). This reduction in ETEC F4 adhesion was also confirmed by using an ex vivo villus adhesion inhibition assay with piglet intestinal villi. As shown in Figure 1, GOS reduced the number of ETEC F4 adhering to the villus epithelium by 55%.
Protecting the gut against ETEC F4 induced intestinal damage
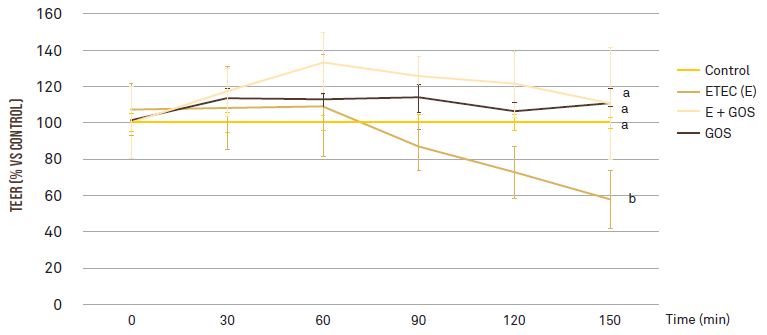
Secondly, the team investigated whether GOS could protect intestinal cells against ETEC F4 induced intestinal barrier injury. Through direct interaction with cell surface receptors, GOS can activate cell signaling that reinforces the epithelial barrier. In this study, cell permeability was assayed by measuring the transepithelial electrical resistance (TEER) and phenol red apparent permeability in Caco-2 cells treated with ETEC F4 and GOS, either alone or in combination. The presence of ETEC F4 induced a significant reduction in the transepithelial electrical resistance and an increase in the permeability of the Caco-2 cells. Nevertheless, this ETEC F4 induced intestinal injury was completely restored when GOS were added (P < 0.001; Figure 2).
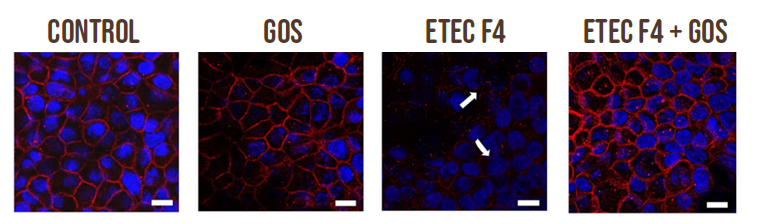
Furthermore, immunolocalization of the tight junction proteins occludin and Zonula Occludens-1 (ZO-1) was evaluated by immunofluorescence in Caco-2 cells treated with ETEC F4 and GOS, either alone or in combination. The presence of ETEC F4 induced occludin and ZO-1 delocalization, indicating cell junction rupture. When ETEC F4 was added in combination with GOS, the tight junction proteins remained correctly localized (Figure 3), which is critical for maintaining epithelial integrity and preventing translocation of pathogens and toxins across the intestinal wall. The positive results on both cell permeability and on tight junction proteins indicate that GOS can prevent ETEC F4 induced intestinal damage.
Inhibiting ETEC F4 induced NF-kB activation
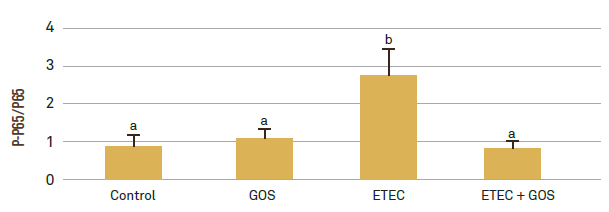
Thirdly, the team investigated whether GOS could protect intestinal cells against ETEC F4 induced intestinal inflammation, as GOS can have direct immunomodulatory properties through interference with cell signaling pathways. The phosphorylated-p65/p65 protein expression ratio was analyzed by Western Blot to measure whether GOS could counteract ETEC F4 induced NF-κB activation leading to inflammation. The p65 protein is a sub-unit of NF-κB, and ETEC F4 induces phosphorylation of the p65 protein, which leads to NF-κB activation. As expected, ETEC F4 significantly increased the protein expression of phosphorylated-p65/p-65, but this was completely counteracted by adding GOS (2.76 vs 0.81, ETEC F4 vs ETEC F4 + GOS, P < 0.05; Figure 4), which indicates that GOS successfully inhibited ETEC F4 induced NF-κB activation.
Conclusion
The Earlyfeed portfolio offers a smart combination of feed solutions, as there is no silver bullet for preventing post-weaning diarrhea. One of the measures taken by Earlyfeed experts is the use of START+, an innovative prebiotic concept based on components that naturally occur in sow milk. Before weaning, START+ stimulates early gut maturation and fosters a healthier microbiome, preparing the piglet ideally for the stressful event of weaning. New research has now also revealed that START+ can play an important role in reducing post-weaning diarrhea by preventing ETEC F4 adhesion and protecting intestinal cells against ETEC F4 induced membrane damage and inflammation. Hence, by mimicking nature’s own protection, START+ can effectively fight post-weaning diarrhea right from the start.
This contact was suggested based on the location you are browsing from. You can of course also consult our other contacts and locations here.